A patent still, also known as a column still or a continuous still, is a type of still used in the distillation of spirits. It is a tall, vertical apparatus with several horizontal plates or trays that act as a series of condensers.
The patent still is the most commonly used type of still in the commercial production of distilled spirits, such as whiskey, vodka, and rum.
The patent still is a very efficient type of still and can produce a very high yield. However, it can also produce a very neutral spirit. To produce a more flavorful spirit, the column still needs to be operated at a lower proof.
The History Of The Patent Still – Coffee Still – Column Still
A column still, also called a continuous still, patent still or Coffey still, is a variety of still consisting of two columns, invented in 1826 by Robert Stein, a Clackmannanshire distiller and first used at the Cameron Bridge Grain Distillery. The design was enhanced and patented in 1831 by an Irishman, Aeneas Coffey.
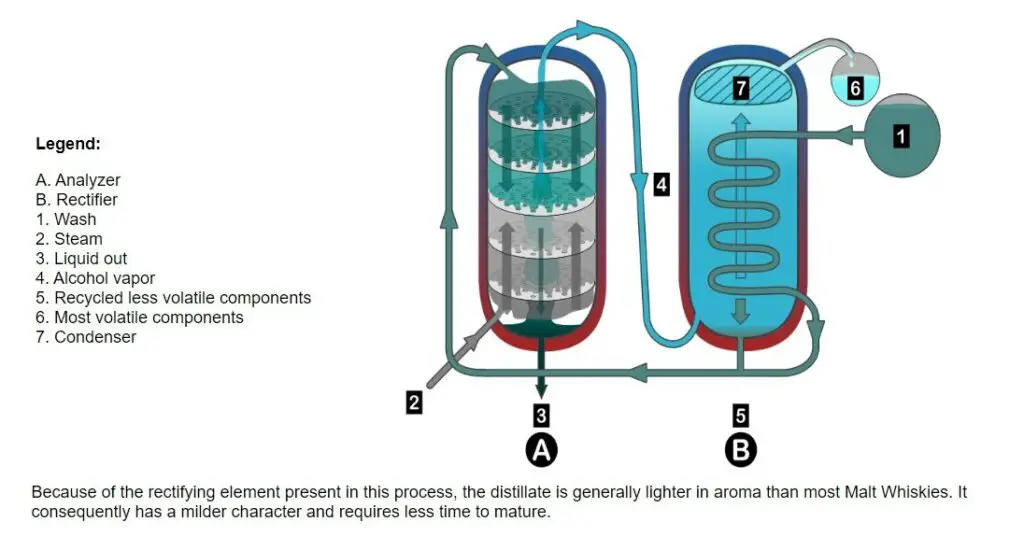
This new still was called the “Continuous Still” (also “Column” or “Patent” or “Coffey” Still). In simple terms, it consists of two columns, one of which has steam rising and wash descending through successive stories inside (referred to as the “Rectifier”).
The steam stripped out the alcohol from the wash and carried over to the second column (referred to as the “Analyzer”) where it circulates until it can condense at the required strength.
How Does A Patent Still Work?
The column is filled with packing material, such as Raschig rings, to increase the surface area with the liquid coming into contact.
The column is heated from the bottom, and the vapors rise to the top. The vapors then condense on the sides of the column and drip down to the tray below. The liquid on the tray closest to the bottom is the purest, as it has undergone the most distillation. The column still is an efficient way to purify liquids, as it can distill a large amount of liquid in a short amount of time.
How To Use A Patent Still (Column Still) To Make Whiskey
The first column (called the analyzer) has steam rising and washing, descending through several levels. The second column (called the rectifier) carries the alcohol from the wash where it circulates until it can condense at the required strength.
Column stills behave like a series of single pot stills, formed in a long vertical tube. The tube is filled with either porous packing or bubble plates. The rising vapor, which is low in alcohol, starts to condense in the cooler, higher level of the column. The temperature of each successively higher stage is slightly lower than the previous stage.
Whereas a single pot still charged with wine might yield a vapor enriched to 40-50% alcohol, a column still can achieve a vapor alcohol content of 96%; an azeotropic mixture of alcohol and water. Further enrichment is only possible by absorbing the remaining water using other means, such as hydrophilic chemicals or azeotropic distillation.
The Column Still Distillation Process
As the boiling whisky vapors travel up through the trays, it cools a bit and droplets of whiskey and water drop out of the vapor and trickle down through the trays to the bottom of the still. The highly concentrated whiskey is taken off the top of the still where it is cooled.
The whiskey vapor from the boiling mash travels up through the bubble caps. The floor of the bubble tray has condensed liquid which the vapor bubbles through. As the vapor bubbles through the liquid some of the vapor cools and condenses. As the liquid builds up in the tray it will overflow the drain pipe and flow down the column to be re-boiled.
A column still is an example of a fractional distillation, in that it yields a narrow fraction of the distillable components. This technique is frequently employed in chemical synthesis; in this case, the component of the still responsible for the separation is a fractionating column.

The Benefits Of Using A Column Still To Make Whiskey
A continuous still can, as its name suggests, sustain a constant process of distillation. This, along with the higher concentration of alcohol in the final distillate, is its main advantage over a pot still, which can only work in batches.
Continuous stills are charged with pre-heated feed liquor at some point in the column. Heat (usually in the form of steam) is supplied to the base of the column. Stripped (alcohol-free) liquid is drawn off at the base, while almost pure alcohol is condensed after migrating to the top of the column.
The Disadvantages Of Using A Column Still To Make Whiskey
The disadvantage of a continuous still, at least for the production of alcohol, is that the alcohol produced is not rendered free of lower-boiling-point contaminants (such as methanol and acetaldehyde).
Today, alternatives open to distillers are to use the labor intensive pot still which carries the fragrances and flavors of the raw material, or the faster, cheaper continuous still with its potential for high strength, pure but tasteless spirit.
What Is a Patent Still? – FAQ
Patent stills are a type of distillation apparatus that are used to purify liquids such as alcohol. The patent still was invented by Scottish scientist and engineer Robert Stein in 1826. Stein’s invention made it possible to distill alcohol on a large scale, and it revolutionized the alcohol industry.
Patent stills work by heating the liquid to be distilled to a vapor, which is then condensed back into a liquid. This process removes impurities from the liquid, leaving behind a pure product. Patent stills are used to produce high-quality alcohols such as whisky, gin, and vodka.
Patent Stills – Conclusion
In sum, a patent still is a common type of distillation apparatus that is used to produce distilled spirits. Patent stills have been in use for centuries, and they are still used today to make a variety of spirits.
If you’re interested in learning more about making your own spirits at home, be sure to check out our blog for more information.
- Vevor Still Instructions - June 12, 2023
- Best Proofing Parrots For Home Distilling - February 1, 2023
- What Is A Porter Beer? - June 12, 2022
